The Muscle Health Research Centre (MHRC) provides a centralized and focused research emphasis on the importance of skeletal muscle to the overall health and well-being of Canadians. Skeletal muscle, 40 per cent of a human's body mass, is a unique and large tissue that significantly contributes to an individual's metabolism, locomotion, and overall quality of life.
Based in the Faculty of Health, the MHRC facilitates the integrated study of muscle biology in the broadest terms, including muscle development, disease, metabolism, blood supply, injury and regeneration, and adaptation to acute and chronic exercise. Approaches used by faculty and graduate students for the study of muscle include molecular, cellular and whole-body techniques.
MHRC Vision
To be Canada's leader in exercise and muscle health research, training and education
MHRC Mission
To achieve this vision via high quality, individual and collaborative research programs, excellent teaching and training of highly qualified personnel in laboratory, technical and communication skills, and the dissemination of research findings to broader communities via publications, seminars and social media platforms.
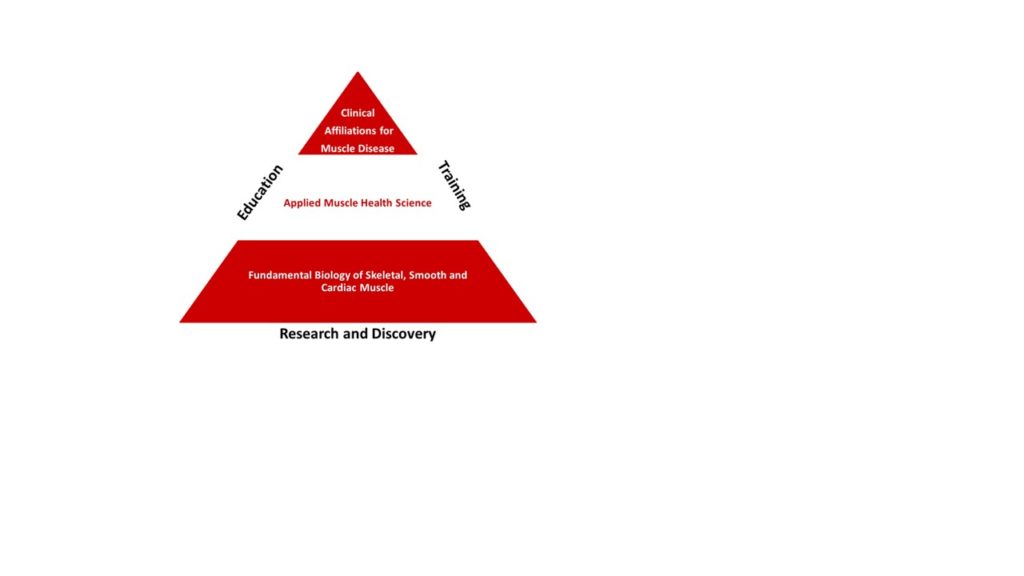
What we do at the Muscle Health Research Centre
What we do at the Muscle Health Research Centre video description transcript
YouTube Playlists
MHRC Seminar Series
Below please find a listing of our Winter 2024 Seminars
Click here to see our complete listing of past MHRC Seminars